After God provided a ram so that Abraham would not have to sacrifice his son, Abraham called that place Jehovah-jireh, which is Hebrew for ‘God is provider’ (Gen. 22:14). A God of love is a God Who provides, not only the requirements needed for life in the first place, but also those things needed for life every day. To assure us that God takes care of our physical needs, Jesus pointed out that God provides for the on-going physical needs of plants and animals (Matt. 6:25-33).
The following article is an excerpt from Devotional Biology: Learning to Worship the Creator of Organisms, Chapter 8.1, pgs. 175-181. The views expressed reflect those of the author, and not necessarily those of New Creation.
God upholds all things (Heb. 1:3), preserves all things (Neh. 9:6; Psa. 36:6), and by Him all things consist and hold together (Col. 1:17). God is responsible for weather1 and the rising and setting of the sun (Psa. 104:19-20; Matt. 5:45). He creates the life of each individual sea animal (Psa. 104:29-30), He is responsible for animal instincts (Job 39:26-30), He makes plants to grow (Psa. 104:14-17; 147:7-8; Matt. 6:28-30), and He provides food for animals of the sea (Psa. 104:25-28), the air (Job 38:41; Psa. 147:7-9; Matt. 6:26), and the land (Job 38:39-40; Psa. 104:21; 147:7-9).
Provision and the Biomatrix
In the last chapter we examined features of the non-biological world that make it possible for organisms to exist. In this chapter we will examine ways in which God designed the biological world to provide for the on-going, daily needs of plants and animals. Animals require food and other nutrients that are not found in the right form in the non-biological world. In the case of the land animals, most of that food and many of the nutrients are provided by plants (Gen. 1:29-30).
However, even the plants need nutrients that are not supplied in usable form by the non-biological world. So, even with all the AP characteristics of the universe in place, plants and animals still could not exist on the earth by themselves. To make life possible for plants and animals, and to further illustrate His attribute of love, God created a complex network of unnoticed organisms (called a biomatrix or organo-substrate)2 that provide what animals and plants need to exist.
Many of the biomatrix organisms are unnoticed because they can only be seen with a microscope: the bacteria (sing. bacterium), most of the algae (sing. alga), and most of the protozoa (sing. protozoan). Other biomatrix organisms (the fungi and the remainder of the protozoa and algae) are large enough to be seen (including the largest organisms on this planet)3, but are rarely seen because they live in places we do not frequent, or appear at times we normally do not observe, or stay inside other things for most or all of their life.
At this point, we know relatively little about the biomatrix. We have only begun to study this long unnoticed part of biology. Let us spend a little bit of time considering this critically important part of the biological world.
Food Providers

All biological organisms need energy to do what they do. Many organisms—including all animals, fungi, protozoa, and some bacteria—get that energy from food (energy-containing molecules formed by other organisms). The energy is first put into organic molecules by producers, organisms specially designed for that purpose. These molecules then become food for all other organisms (the consumers). Organisms of the biomatrix play a very important role in providing food to many of the animals of the world, especially those in water environments.
Producers in Water Environments
The main producers on the land are plants.4 All other producers (the algae and some of the bacteria) are part of the earth’s biomatrix. Some algae5 and photosynthetic bacteria produce on the land what the plants do not. The remainder of the algae and photosynthetic bacteria are the main producers for the streams, lakes, and oceans of the world. There are thousands of species of algae and thousands of species of photosynthetic bacteria in an amazing variety of forms—a vast percentage of which are microscopic.6
As in the case of plants, algae and photosynthetic bacteria construct organic molecules by the process of photosynthesis (Greek words phōs, ‘light’, + synthesis, ‘putting together’). One group of algae, the green algae, are green because they collect energy from the same colors of sunlight that plants do. Other algae and photosynthetic bacteria use different pigments to absorb different colors of sunlight. Consequently, they display different colors and are often named accordingly (e.g. ‘red algae’, ‘brown algae’, ‘golden algae’, ‘yellow-green algae’, ‘cyanobacteria’, ‘purple bacteria’).
Producers in Extreme Environments
Plants, algae, and photosynthetic bacteria produce almost all the food needed by the consumers of the world, but not quite all. There are some environments where plants, algae and photosynthetic bacteria cannot live, and where their energy-containing organic molecules cannot reach. Algae and photosynthetic bacteria, for example, cannot perform photosynthesis without light, so cannot live their entire life in the soil or in rocks, inside organisms, or in deep water where light cannot penetrate. They also cannot live in ‘extreme’ environments—namely wherever there is too little water or where there are too few nutrients, or wherever temperatures are too hot or too cold, or wherever the pH is too high or too low, or wherever the salinity is too high, or wherever the pressure is too high.

All the producers of these extreme environments are members of the biomatrix. Lichens, for example, are the main producers in land environments where nutrients are hard to find or water is in short supply. The fungi that make up lichens are capable of absorbing nutrients when there are not enough of them to support plants, and capable of pulling water out of the air—even the dry air of deserts. The algae or photosynthetic bacteria that cooperate with the fungi to make up the lichens produce the energy containing organic molecules needed by both organisms. The producers in most other extreme environments are bacteria—each specially designed to live in a particular kind of extreme environment.
In extreme environments where light is absent, producers construct their molecules by the process of chemosynthesis. Rather than using light energy, chemosynthesis uses ‘chemo’, or chemical energy, to synthesize energy-containing organic molecules. Around hot springs on the bottom of the ocean, for example, chemosynthesizing bacteria are producers for a fairly diverse group of animals, including tube worms, shrimp, crabs, fish, and octopuses. Chemosynthesizing bacteria often inhabit environments where no other organism can survive.
In Yellowstone National Park, for example, the waters flowing out of hot springs support a fairly diverse community of biomatrix organisms, creating, in turn, the beautiful colors seen in Yellowstone hot springs. In the extremely hot water at the source of the springs, the producers are chemosynthesizing bacteria that thrive in hot, nearboiling water. In the progressively cooler waters found at progressively greater distances from the springs, the producers are photosynthesizing bacteria and algae.
Other chemosynthesizing bacteria are designed to thrive in very acidic (low pH) environments. Others still live in very alkaline (high pH) environments. Others live in very salty environments—some planted directly on crystals of salt! Still others live miles beneath the surface under the extreme pressures that are caused by miles of overlying rock.
First consumers
Plants come in such a wide variety of sizes that nearly every size of land animal has a suite of plants that they can consume directly. Most of the biomatrix producers, on the other hand, are too small to directly provide food for animals. For example, most of the animals of the water environments of the world are fed actually fed by protozoa. Protozoa are biomatrix consumers that eat the tiny food particles produced by smaller bacterial producers, and in turn, become the food for progressively larger animals.
Break Down Molecules
Producers use energy from their environment to build energy-containing organic molecules. The energy stored in these molecules can be extracted by breaking them back down again. In fact, the breaking down of energy-containing organic molecules is how consumers get their energy, including many of the organisms of the biomatrix.
Digestion
Animals must consume food (energy-containing molecules) to survive. Animals digest the food by breaking down molecules into simpler molecules. However, not all the food that animals consume can actually be digested by those animals. To help animals with digestion, an entire community of bacteria and fungi inhabit the digestive systems of animals—perhaps all animals. It is this gut flora7 that is largely responsible for the digestion of grass in ruminants like cattle and for the digestion of wood in termites.
The gut flora in humans may involve hundreds of species and include 100 trillion individuals—more than ten times the total number of human cells in the human body. Without biomatrix organisms that help in digestion, most animals would not be able to survive.
Decomposers

When an organism dies, it is important that the body of that organism be broken down (decomposed) into the simple molecules from which it was constructed. There are a couple reasons for this. First, if bodies were not decomposed, we would quickly find ourselves walking on a very deep pile of dead bodies. Second, if organic molecules were not returned to the molecules from which they were made, the earth would run out of these molecules.
Decomposition cleans the earth of dead bodies and dangerous wastes and replenishes the supply of nutrients needed by the earth’s organisms. Almost all decomposition is done by bacteria and fungi of the earth’s biomatrix. Without biomatrix decomposers the earth would be polluted with dead bodies and waste, and organisms would run out of nutrients.
Convert Molecules
Fixation
A number of elements are needed by organisms (e.g. oxygen, carbon, hydrogen, nitrogen, calcium, phosphorus, potassium, sulfur, sodium, chlorine, magnesium, boron, cobalt, copper, fluorine, iodine, iron, manganese, molybdenum, selenium, silicon, zinc). Outside of organisms, each of these elements is available in one or more inorganic forms.
The most common elements of life are found in the atmosphere: oxygen in the form of oxygen gas (O2), water in the form of water vapor (H2O), carbon in the form of carbon dioxide (CO2), methane (CH4), hydrogen in the form of hydrogen gas (H2), and nitrogen in the form of nitrogen gas (N2). Most of the remaining elements are found in minerals in rocks. In the case of most of these elements, the natural, inorganic form is not in a form that can be easily used by organisms. Most of the molecules have to be broken apart to release the needed elements, and some of the bonds are extremely difficult to break. In the case of several of the elements, even the atoms have to be altered by taking away or adding electrons.
If the inorganic form of an element has to be substantially changed for it to be usable by organisms, it is said that the element must be fixed. The process that accomplishes the ‘fixing’ of that element is called fixation. The element carbon, for example, must be fixed to take it out of carbon dioxide and construct organic molecules from it. Almost every organic molecule is storing more energy in its bonds than carbon dioxide, so building organic molecules from carbon dioxide is a rather difficult process that requires considerable energy. Certain organisms had to be provided with rather complex designs in order to fix carbon. In plants, algae, and photosynthetic bacteria, for example, carbon fixation has been designed as part of the process of photosynthesis. In fact, carbon fixation is a process God designed into all producers, whether they build their organic compounds by photosynthesis or chemosynthesis. Consequently, much of the carbon fixation that occurs on the earth is performed by algae and bacteria of the biomatrix.
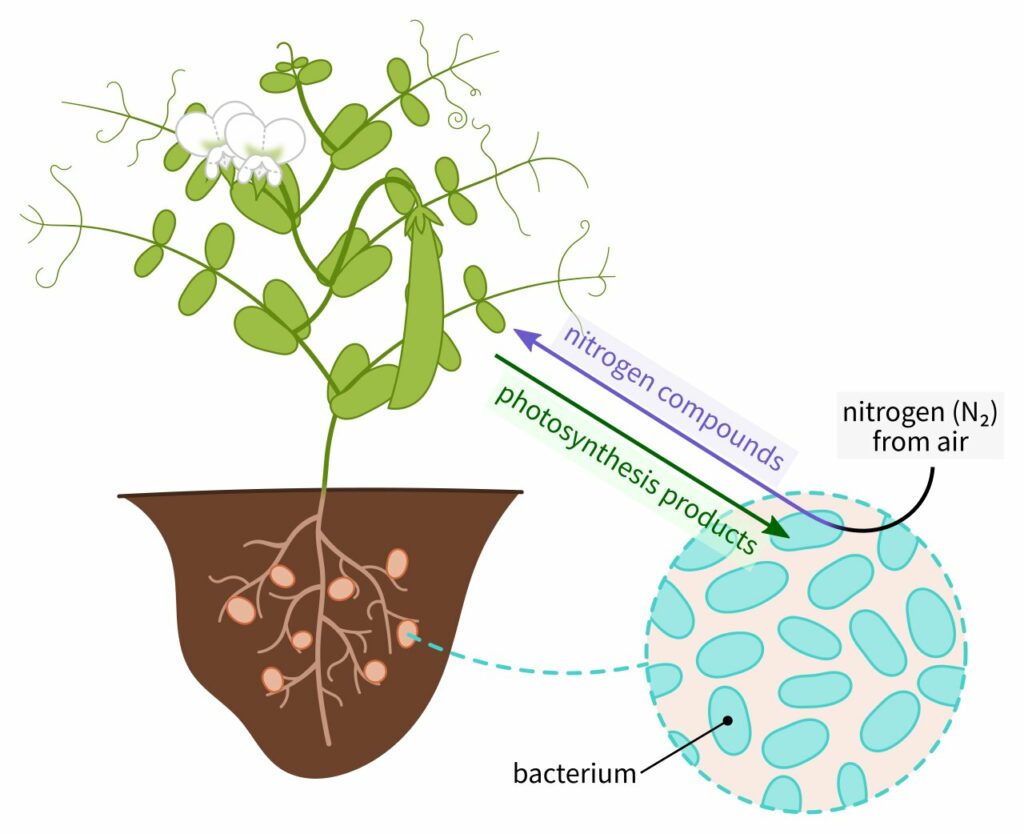
Another life-essential element that requires fixation is nitrogen. With 81% of the earth’s atmosphere made up of nitrogen gas, nearly every organism on earth has easy access to inorganic nitrogen. Very few organisms, however, can use this form of nitrogen. Each particle (molecule) of nitrogen gas is made up of two atoms of nitrogen bound together by one of the strongest chemical bonds known. Because it is very difficult to break that bond, nitrogen gas does not enter into very many chemical reactions.
On the one hand, this makes nitrogen gas a great choice for making up most of the atmosphere. On the other hand, it is difficult to separate the nitrogen atoms so that they can be used by organisms. Extremely hot fires and billion-volt lightning bolts are some of the very few things outside organisms that can break these bonds. To make nitrogen available to organisms, nitrogen must be fixed. The only organisms capable of nitrogen fixation are some bacteria in the biomatrix known as nitrogen-fixing bacteria.
The inorganic form of a majority of the life-essential elements is available only in rocks. In several cases (such as for molybdenum) the element is found in very low concentrations, even in rocks where it is most abundant. In rocks, the element is often available in parts per thousand or parts per million. For any of these elements to be used by organisms, it must somehow be mined from the rocks.
In the case of a couple of the elements, bacteria have been discovered that mine that element from the rock. In each case a special type of bacterium living in water releases a molecule into the water that ‘lands’ on the rock surface and bores into the rock until it encounters an atom of that element. It then jettisons a portion of the molecule attached to the element into the water to be picked up by the mining bacterium. It is likely that for each of the elements necessary for life that is found in minerals, there is at least one bacterium species of the biomatrix that is designed to mine that element from rock and fix it for use by organisms.
Remediators
Bacteria of the earth’s biomatrix also work at keeping the earth safe for life. There are times when organisms release waste that is unhealthy for other organisms. There are other times when non-biological processes like volcanoes, hot springs, and storms expose organisms to dangerous substances that have been buried in the rocks of the earth. Our experience is that if we wait long enough these dangerous substances disappear. Closer examination usually reveals that there is a particular bacterium that transforms that dangerous substance into something that is not hazardous to other organisms.
It would appear that for each dangerous substance that might appear in the world, God designed a bacterium to remediate—to take the toxicity out of dangerous molecules. Thus, some of the biomatrix bacteria have been created for the purpose of bioremediation. As in the case with other aspects of the biomatrix, we are only just beginning to learn about bioremediation. New bacteria are being discovered all the time that make toxins non-toxic, place radioactive elements in safer molecules, etc.
Provide Nutrients
Develop Soil

“Every spoonful of garden soil contains some 1010 bacteria…”8—that is 10 billion bacteria! What many of those bacteria are doing—in a non-stop fashion, 24 hours a day, 7 days a week, 365 days a year—is generating nutrients that plants need to survive. Fixers are putting needed elements into forms the plants can use, decomposers are breaking down dead plant and animal material for re-use by the plants, and bioremediators are making dangerous molecules safe.
Most plants require soil to thrive and biomatrix bacteria are mostly responsible for making soil ‘fertile’. That is why when a sterile landscape is formed—such as after a very hot fire or after lava flows across the earth’s surface—bacteria are the very first organisms to inhabit that landscape. They immediately begin the process of building soil so that plants can eventually thrive in that area.
Feed and Water Plants
The role of the biomatrix organisms does not end with creating the nutrients. Some organisms of the biomatrix also deliver the nutrients. In the case of trees, for example, studies indicate that tree roots alone are not capable of absorbing water fast enough to supply the needs of the tree. Closer examination reveals that the roots of the tree are not alone. Fungi have been discovered to be associated with the roots to help in absorbing the needed water and nutrients. These fungi are called mycorrhizal fungi (Gk. mykós, ‘fungus’ + riza, ‘roots’). Fungi have been specially designed by God to be more effective at pulling substances into their bodies than any other kind of organism.
Combined with the absorbing ability of the plant roots, mycorrhyzal fungi can, in fact, absorb enough water and nutrients to supply the needs of even the very largest trees. Research suggests that these fungi may be used by all woody species of plants, and perhaps by a vast majority of all plants.
Vitamin Source
Biomatrix bacteria also produce vitamins that are necessary for the health of plants and animals. Vitamin B12, for example, cannot be produced by any animal or plant—it can only be produced by biomatrix bacteria. In humans, bacteria in the large intestine produce Vitamin K and Vitamin B7 (also known as biotin or vitamin H).
Protection
To keep them healthy after the Fall, God designed organisms with rather complex systems of protection from pathogens. Part of this involves keeping the pathogens from getting into an organism’s body. This includes surrounding organisms with tough coverings that are hard for pathogens to get through. But it also includes planting friendly microorganisms on the outside of those tough coverings so that pathogens are prevented from living on those coverings. Being microscopic, these friendly microorganisms are part of the biomatrix, and created to protect larger organisms. Humans, for example, have a skin flora designed to provide our first line of defense against disease.
“Consider the lilies of the field… even Solomon in all his glory was not arrayed like one of these… if God so clothes the grass of the field… will He not much more clothe you…?”
Matt. 6:28-30, NKJV
Learn More about the Biomatrix
Footnotes
- God is responsible for springs (Psa. 104:10) as well as the evaporation of water (Psa. 135:5-7), the formation of clouds (Job 37:10-11; Psa. 147:7-8), thunder & lightning (Job 37:5; Psa. 135:5-7), wind (Psa. 135:5-7), and the dropping of frost (Job 37:10), snow (Job 37:5-6) and rain (Job 5:10; 37:5-6; 38:36; Psa. 147:7-8; Matt. 5:45). ↩︎
- The word ‘biomatrix’, suggested by Joseph Francis, is derived from the Greek word bios meaning life (both because it is necessary for animal (and plant) life and because it is made up of biologically living organisms) and ‘matrix’ (both because it is a rather complex system of organisms and because it is an unnoticed basis of the reality—as the ‘matrix’ was, as in the 1999 American-Australian science fiction action film ‘the Matrix’). ‘Organo-substrate’ is derived from ‘organic’ (both because it is part of carbon-based or organic life and because it is necessary for the existence of carbon-based plant and animal organisms) and ‘substrate’ (because it is the substrate or ground or foundation for animal and plant life). ↩︎
- Some fungi in the soil and some algae in the sea are spread over square miles of earth surface, and are in the running for the largest individual organisms on the planet. ↩︎
- Although nearly all plants are producers, there are a very few plants that are not (such as the Indian pipe, Monotropa uniflora). ↩︎
- As in the case of plants, although nearly all algae are producers, a few species are not. ↩︎
- All bacteria, and all but a few species of algae (e.g. the kelp and seaweeds) are microscopic. ↩︎
- When first discovered, microscopic organisms were classified among the ‘plants’. The plants living in a particular area were known as the ‘flora’ of that area, so the microorganisms of the stomach and intestines of animals were referred to as the ‘flora’ of the gut, or ‘gut flora’. ↩︎
- Margulis, Lynn, and Karlene V. Shwartz, 1998, Five Kingdoms: An Illustrated Guide to the Phyla of Life on Earth, Third Edition, W. H. Freeman, New York, NY, 520 p. (p. 44). ↩︎